AbCellera Biologics (ABCL)·Q4 2025 Earnings Summary
AbCellera Crushes Q4 on $45M Patent Settlement, Stock Surges 6%
February 24, 2026 · by Fintool AI Agent

AbCellera Biologics (NASDAQ: ABCL) reported Q4 2025 results today that crushed expectations, driven by a one-time patent litigation settlement. Revenue of $44.9 million vastly exceeded the $6.3 million consensus, while the net loss of $0.03 per share was far better than the expected $0.16 loss. Shares rose 6.3% to $3.22 in regular trading and climbed another 5% after hours to $3.37.
The quarter's performance was dominated by a $45 million upfront settlement payment from patent litigation, which accounted for 60% of full-year 2025 revenue. Excluding this one-time item, the underlying business continued its transition to a clinical-stage biotech with two programs now in clinical trials.
Did AbCellera Beat Earnings?
Massive beat on both revenue and EPS, though heavily influenced by non-recurring items.
Full Year 2025 Summary:
How Did the Stock React?
ABCL shares surged on the earnings beat:
The stock remains well below its 52-week high of $6.52 and 200-day moving average of $3.96, reflecting ongoing skepticism about the biotech's path to profitability. Today's move represents the largest single-day gain since Q2 2025 earnings in August.
Earnings Reaction History:
What's Driving the Revenue Spike?
The Q4 revenue surge was almost entirely driven by a one-time patent settlement:
Revenue Composition (Q4 2025):
- Licensing & Royalty Revenue: $46.9M (includes $45M settlement)
- Research Fees: $27.2M (full year)
- Milestone Payments: $1.0M
CEO Carl Hansen framed the results positively: "In 2025, AbCellera successfully delivered on all its corporate priorities, transitioned to a clinical-stage biotech company, and ended the year with approximately $700 million in available liquidity to execute on our strategy."
Without the settlement, Q4 operating revenue would have been roughly flat to prior quarters. The core partnership business continues to generate research fees, with 8 new partner-initiated programs started in 2025 (bringing cumulative total to 104 programs with downstream participation).
What Changed From Last Quarter?
Key developments since Q3 2025:
The most significant development was ABCL635 advancing to Phase 2 at year-end 2025, with the Phase 2 portion officially initiated in January 2026.
What's the Pipeline Status?
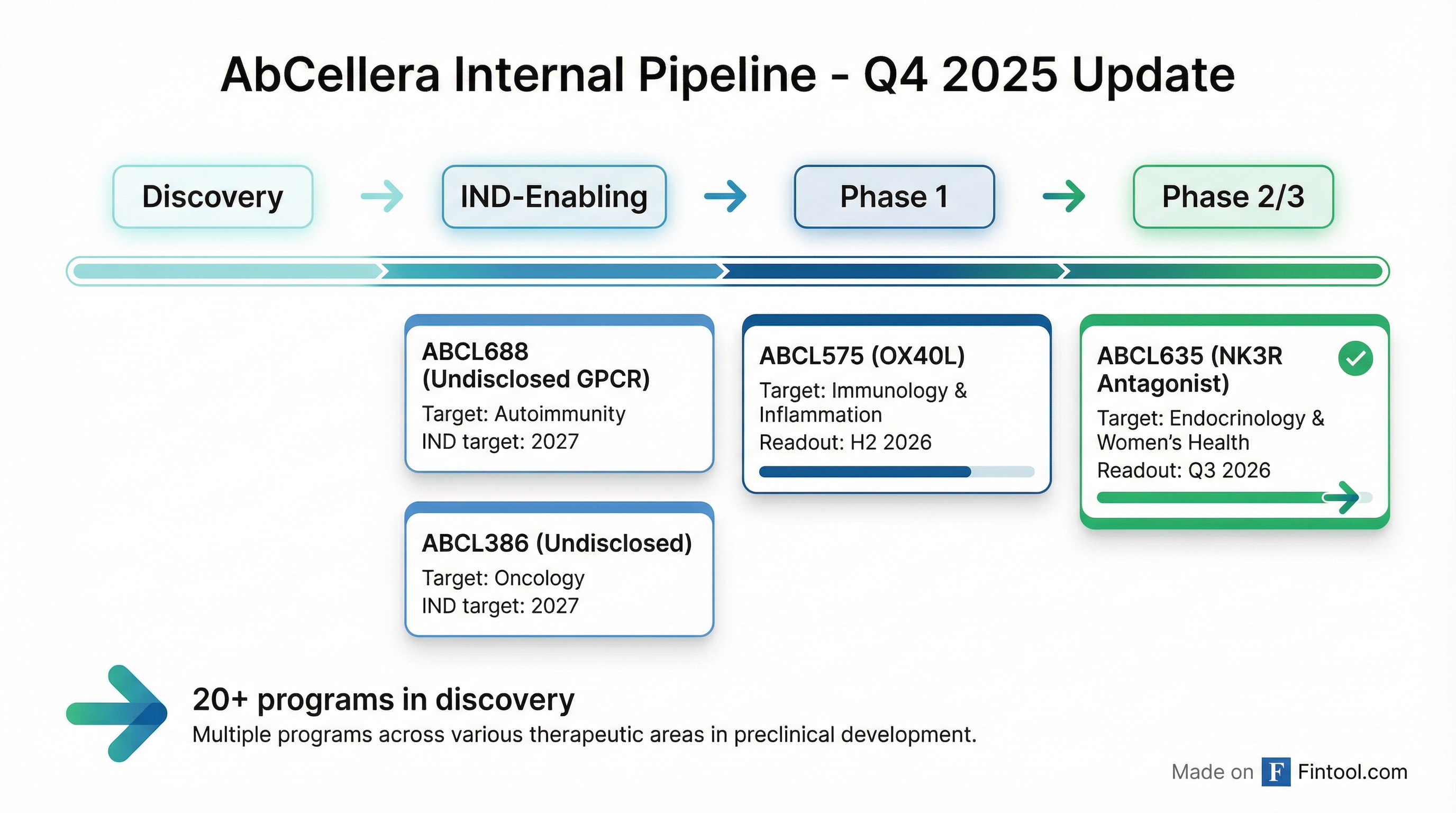
AbCellera now has 4 internal development candidates and 20+ programs in discovery:
ABCL635 Target Product Profile:
- Once monthly subcutaneous self-injection
- Differentiated safety profile vs small molecules
- Comparable efficacy to approved treatments
What Did Management Guide?
AbCellera did not provide explicit financial guidance but outlined 2026 priorities:
- ABCL635: Phase 1/2 topline readout in H2 2026
- ABCL575: Phase 1 topline readout in H2 2026
- ABCL688: Progress through IND-enabling studies
- ABCL386: Progress through IND-enabling studies
- Pipeline: Nominate at least 1 additional development candidate
Liquidity Position:
Management stated this provides sufficient capital to fund "well beyond the next three years of pipeline investments."
What About the Partner Business?
The antibody discovery platform continues generating partnership activity:
Partner Program Status (of 104 total):
- In progress or AbCellera leading: 90 programs
- Clinical development: 6 programs
- Preclinical development: 84 programs
- Not expected to progress: 14 programs
Notable partner updates this quarter:
- Arsenal Bio's AB-3028: IND authorized
- Dechra/Invetx program: Pivotal studies initiated
What Are the Key Risks?
Near-term concerns:
- Revenue sustainability: Q4 revenue was 95% one-time settlement; underlying quarterly run-rate remains ~$5-10M
- Clinical execution: Two Phase 1 readouts in H2 2026 represent binary risk events
- Cash burn: Operating expenses of $292M annually against minimal recurring revenue
- Partner concentration: Research fee revenue depends on pharma R&D budgets
Balance sheet highlights (Dec 31, 2025):
What Should Investors Watch?
2026 Catalysts:
2027 Potential Catalysts:
- Late-stage development of ABCL635 in menopausal VMS
- Phase 2 initiation of ABCL635 in oncology VMS
- Out-licensing options for ABCL575
- 3 IND submissions (ABCL688, ABCL386, new candidate)
Bottom Line
AbCellera delivered a headline-grabbing Q4 with revenue 6x above estimates, but the underlying story is the transition to clinical-stage biotech rather than financial performance. The $45M patent settlement masks a business still burning ~$130M annually on operations.
What matters for 2026:
- H2 readouts for ABCL635 and ABCL575 will determine whether the internal pipeline thesis is working
- $700M liquidity provides runway, but investors will watch cash burn vs milestone achievement
- Partner business continues steady progress toward potential future royalties
The stock's 6% move reflects optimism about the balance sheet and pipeline progress, but with shares still 50% below the 52-week high, the market awaits clinical proof points before re-rating the story.
View AbCellera Company Profile · Read Q4 2025 Earnings Transcript · View Prior Quarter (Q3 2025)